Given the abundance of electronic devices around us, it is challenging to envision modern life without batteries. These gadgets rely on different types of batteries for power. As efficient energy storage solutions, batteries are widely used in various applications. They play an inevitable role in the shift toward sustainable energy. Since the mid-18th century, battery designs have undergone significant changes continuously, driven by continuous technological progress. The electrolytes used in batteries have evolved from liquid or gel-based forms to solid-state materials. The current research to bring solid-state battery technology to market has made remarkable progress recently. The primary concern in developing new eco-friendly and sustainable green energy-based battery technologies is to cut down greenhouse gas emission and reduce the effects of global warming on climate change.
STANDARD CELLS
The journey of energy storage devices started with the study by Italian physicist Alessandro Volta, who generated the steady electric current with the Voltaic Pile (the first electrical battery) during 1799-1800. A battery is an energy storage device that stores chemical energy and converts it to electrical energy via electrochemical reactions. Thus, it is used as an energy source. A battery consists of one or more electrochemical cells. It has two electrodes (terminals) — positive (a cathode) and negative (an anode). In simple terms, it is a portable power bank that releases electricity or electric current when required. The flow of current between electrodes is mediated by electrolyte. Basically, electrolytes are media responsible for ionic conduction, i.e., ion transfer (positive and negative) between electrodes while charging and discharging. Different electrolytes (liquid or gel) were used in a variety of cells (viz., Zinc-Carbon batteries, Alkaline batteries, Lead-Acid batteries, Nickel-Cadmium batteries) prior to Lithium-ion batteries. For instance, dilute sulphuric acid (a liquid) is used in Lead-Acid batteries (also known as car batteries). One can immediately figure out the use of heavy and toxic materials (like Lead and Cadmium), which pose serious concerns and may be environmentally hazardous.
There are two types of batteries: primary (disposable) and secondary (non-disposable). In primary batteries (viz. zinc-carbon battery), like what we use in torches and other portable devices, the material is consumed by chemical reactions. Thus, such a battery can be used only once because it cannot be recharged since the electrochemical reactions in this case are irreversible. On the other hand, secondary batteries can be operated by discharging and recharging, i.e., they are rechargeable. In fact, the reversible electrochemical reactions can restore the active materials of such batteries. The re-usability of secondary batteries with high power densities and discharge rates makes them advantageous over primary batteries.
LITHIUM ERA
We all know that batteries used in laptops, smartphones, and electric vehicles are Lithium-ion (Li-ion) batteries. They are all around us to power the modern world. The Li-ion batteries are rechargeable that use Li ions to move between the cathode and the anode while charging and discharging. These are quite popular because of their high energy density and long cycle life. The high energy density means that the Li-ion batteries can store much energy in a small space. They are lighter, charge faster, and last longer than traditional batteries. In addition to Li-ion batteries, the development of new rechargeable batteries like metal-hydride batteries, nickel-cadmium batteries, and sodium-ion batteries has also taken place.
Though these batteries have played a crucial role in the rise of electric vehicles, the safety concerns, mainly the possibility of catching fire immediately, cannot be overruled in case they are overheated or damaged by any means. Another serious concern is related to the environment.
The mining process for elements like lithium, cobalt, nickel, etc., might be harmful to the ecosystem, and recycling old Lithium-ion batteries is also not easy. For greater safety and battery performance, experiments with solid-state batteries are in news with exciting results. The Li-ion batteries are the primary driving force behind the development of the modern tech world for a sustainable and brighter future.
THE SOLID STATE SURGE
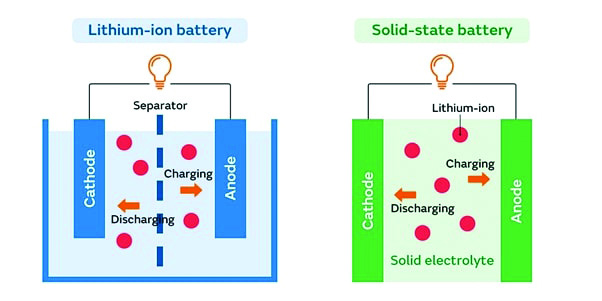
The technology concerning Li-ion batteries has been developed and widely used for many years. However, the invention of solid-state batteries (SSBs) has great potential to revolutionise our way of storing and using energy for different purposes. In SSBs, one uses a solid electrolyte to transfer ions between electrodes, unlike Li-ion batteries, where we use liquid electrolytes. These electrolytes may be ceramics and polymers. Using solid electrolytes leads to several advantages over liquid electrolytes in traditional batteries. SSBs not only perform with higher energy densities, but also have a longer lifetime. These batteries are safer as well as environmentally friendly. Further, their faster charging rate under certain conditions, compared to traditional batteries, makes them suitable for wider applications. Indeed, the most significant advantage of SSBs is that they can hold more energy at the same size as a Li-ion battery. In addition, replacing liquid with a solid electrolyte prevents SSBs from catching fire since the flammable components are eliminated. The SSBs seem to electrify our tomorrow because they are compact (i.e., lighter and thinner in size) and have far better higher energy density, safety standards, life, and charging potential.
However, there are several challenges in making SSBs. The interface issue, i.e., the poor contact between the electrodes and the solid electrolyte used, is a problem that reduces the ionic conductivity. Further, it is also difficult to process ceramics and prepare them as electrolytes since they are brittle. Temperature sensitivity is another issue to deal with, since room temperature performance in the case of some solid electrolytes has yet to be established. Moreover, the new production techniques are also required to scale up such batteries from the laboratory to the market.
CONCLUSION
The SSBs are new and have the potential to replace the Li-ion batteries in the future. The SSBs are still in the development phase with many ongoing experiments. Various research groups with industry partners are working to solve the problems involved so that SSBs can be ready for use in the coming days. The emergence of SSBs is expected to revolutionise grid energy storage due to their higher capacity and longer life cycle. These batteries might also be helpful in renewable energy integration, especially in the case of solar and wind energy, which are intermittent renewable energy sources provided by nature.
*The writer is a Professor of Physics and Director, Research & Development Cell, HNBGU, Srinagar-Garhwal, Uttarakhand. He can be reached at hemwati.nandan@hnbgu.ac.in.










