TECH TALK / GLOWCAS9
For decades, gene therapy has held the promise of curing some of humanity’s most devastating hereditary diseases. Yet, while CRISPR-Cas9 revolutionised genome editing by making DNA cuts with extraordinary precision, one major limitation persisted: scientists could not observe the Cas9 enzyme at work inside a living cell. Traditional methods required breaking open or fixing cells destroying the system they hoped to study. Real-time tracking remained out of reach.
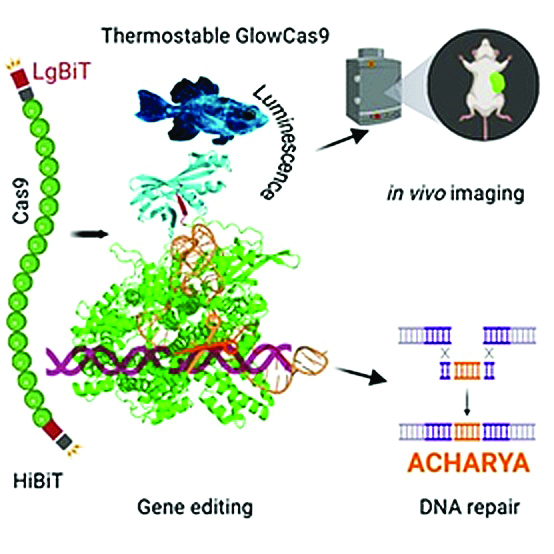
A team at the Bose Institute, Kolkata, led by Dr Basudeb Maji, has now solved this long-standing problem by developing GlowCas9, a bioluminescent version of Cas9 that produces light when active. Their findings, published in Angewandte Chemie International Edition, represent a significant leap forward in both fundamental biology and applied genetic therapy.
GlowCas9 was designed by PhD researcher Arkadeep Karmakar, who fused Cas9 with two inactive halves of a split nano-luciferase enzyme. This luciferase, originally derived from deep-sea shrimp proteins, only lights up when its two pieces come close enough to reassemble. When Cas9 folds correctly and initiates gene editing, the luciferase fragments reunite and emit a glow—a molecular signature that marks the precise timing and location of DNA cutting. The effect is subtle but powerful: a measurable burst of light that allows scientists to track CRISPR activity without harming the cell.
This bioluminescent behaviour solves a fundamental challenge in genome engineering. For the first time, researchers can watch the ‘molecular scissors’ at work, cutting DNA, engaging repair pathways, and completing edits, in living cells, tissues, and even intact plant leaves. The ability to visualise the process as it happens opens new possibilities in understanding CRISPR’s efficiency, its stability inside complex biological environments, and the timing of DNA repair mechanisms.
GlowCas9 is not merely a visualisation tool. Early studies show that it is more stable than conventional Cas9, maintaining structure and activity even at higher temperatures. This is important for therapeutic applications, where enzymes must remain robust inside the body. The researchers also found that GlowCas9 improves the efficiency of homology-directed repair (HDR), the high-precision DNA repair mechanism used to correct mutations responsible for diseases like sickle-cell anaemia and certain muscular dystrophies.
In a symbolic demonstration of its accuracy, the team programmed GlowCas9 to insert the DNA sequence for ‘ACHARYA’ into a genome— an homage to Acharya Jagadis Chandra Bose, the institute’s founder and one of India’s pioneering scientists. This precise, efficient insertion highlights the tool’s potential in corrective gene therapy, where targeted edits must be carried out without introducing off-target damage.
Another promising application lies in agriculture. Because GlowCas9 can be visualised in plant systems without introducing foreign DNA, it may facilitate non-transgenic gene editing in crops, offering safer and more publicly acceptable avenues for improving plant traits.
GlowCas9 also represents a major step toward what scientists call ‘thera tracking’, the ability to simultaneously deliver gene therapy and track its success in real time. Current CRISPR-based therapies often rely on delayed or indirect markers to confirm whether a gene edit was successful. With GlowCas9-like systems, clinicians could one day map when Cas9 reaches the target tissue, observe when it cuts DNA, and monitor whether repair proceeds correctly. This would dramatically improve safety in clinical trials and personalisation of treatments.
More broadly, GlowCas9 highlights India’s growing contributions to frontier biotechnology. As global research groups race to develop safer, more controllable gene-editing platforms, the Bose Institute’s innovation positions Indian scientists at the forefront of next-generation genetic tools. By merging gene editing with light, GlowCas9 turns a once-hidden process into something observable, measurable, and ultimately more controllable. In doing so, it brings gene therapy closer to a future where doctors can not only correct faulty genes but literally watch healing begin.