NEW FRONTIERS OF SCIENCE
Every innovation begins as a quiet possibility. A researcher notices an unexpected laboratory result, an engineer identifies a long-standing societal problem, or a student imagines a better way to complete a routine task. At this earliest stage, nothing is written, tested, or demonstrated. There is only thought. This informal beginning, sometimes called TRL 0 (Technology Readiness Level), is where most ideas remain, and that is natural. Imagination must always exceed what is built, for it is the source of originality. Yet when a particular idea refuses to fade, it must begin the long journey from possibility to proof, and from proof to practice.
That journey is rarely linear. It loops through setbacks, redesigns, failures, renewed attempts, and slow realisations. To navigate this uncertainty, NASA introduced the Technology Readiness Level framework in the 1970s; in the 1990s, John C Mankins formalised its definitions and evidence expectations. TRLs have since travelled far beyond space missions. They are now used in energy, water, agriculture, and healthcare to give innovators a shared vocabulary and to anchor maturity claims to evidence gathered under increasingly realistic conditions. Instead of vague statements about progress, TRLs offer a clear map from insight to impact.
THE TRL JOURNEY
TRL 1 begins with scientific observation. Imagine a materials scientist who discovers that a certain natural surface can adsorb heavy metals from contaminated groundwater. At TRL 2, the idea becomes a concept. The scientist imagines a low-cost water purification device, writes down hypotheses, and sketches possible configurations. TRL 3 demands proof of concept through controlled experiments. The material is tested repeatedly to confirm its behaviour. If results are consistent, confidence grows.
TRL 4 builds a prototype and tests it in controlled laboratory conditions. These early stages can feel comfortable, as conditions are controlled and progress is legible to peers. Research papers are written and prototypes are demonstrated, yet the device still exists in an environment that behaves perfectly. As many innovators admit, TRL 1 to TRL 4 are also the stages where technology remains in the laboratory. The purifier works in glass beakers and polished rigs, but life outside the laboratory is untidy, imperfect, and unpredictable.
TRL 5 marks a turning point. The prototype must now perform in relevant environments. Real groundwater carries unpredictable impurities, suspended solids, and fluctuating pH. TRL 6 requires pilot testing in the field. A small unit is installed in a community, where researchers observe flow rates, clogging, maintenance needs, and user interactions over several days or weeks. TRL 7 demands sustained performance in real settings. The purifier must tolerate inconsistent cleaning, irregular usage, seasonal variation, and unpredictable behaviours. When problems arise, the design must adapt, because reality does not follow laboratory schedules.
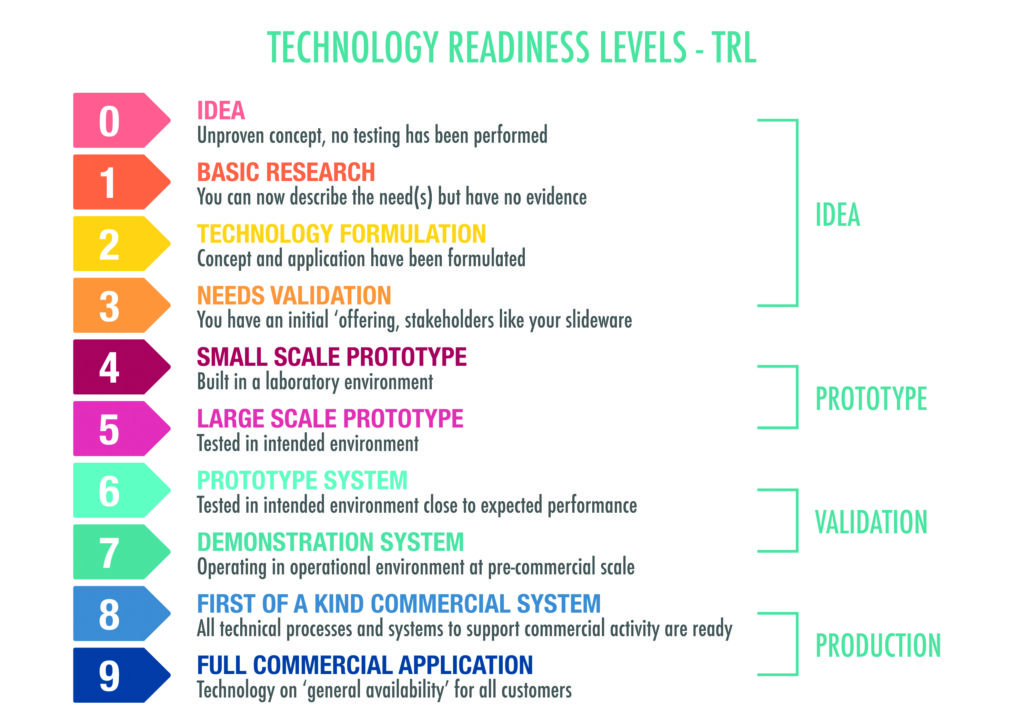
If the system endures these trials, it reaches TRL 8, where it is considered mature and ready for deployment at scale. TRL 9 represents full integration into daily life. Many filtration systems and disinfection units used in low-resource settings travelled this path over years before earning widespread trust. A parallel journey unfolds in healthcare. A point-of-care diagnostic begins with biomarker discovery at TRL 1, reaches prototype validation at TRL 4, enters clinical testing at TRLs 6 and 7, and only reaches TRL 9 after regulatory approval, manufacturing readiness, supply chain preparation, and training for frontline workers. Rapid diagnostic kits for infectious diseases, now used widely across continents, followed this long arc.
Despite such successes, most innovations never reach TRL 9. Studies frequently describe a valley of death between TRL 4 and TRL 7, where promising technologies collapse despite scientific merit. One example is a low-cost arsenic filter that worked in laboratories and early pilots but failed during scale-up because maintenance behaviour was misunderstood, supply chains for consumables were thin, and willingness to pay was overestimated. In diagnostics, validated devices stall when certification is slow, distribution networks are weak, or health systems are unprepared to adopt new workflows. These cases show that technical readiness is necessary but not sufficient.
Field observations reinforce this lesson. During neutral, observation-driven pilots, teams may find that households skip periodic cleaning to save time, which undermines performance. In another setting, spare parts are unavailable locally, so devices sit idle after a minor failure. Elsewhere, water chemistry changes between seasons, saturating filter media faster than expected. Such outcomes confirm why TRL 6 and TRL 7 must include behavioural and logistical evidence, not only technical metrics. Funding calls increasingly require a stated TRL with traceable evidence, a practice formalised in European programmes where target TRLs are specified. This creates comparability but also demands honesty and rigour from innovators.
BEYOND TECHNOLOGY: THE IRL LENS
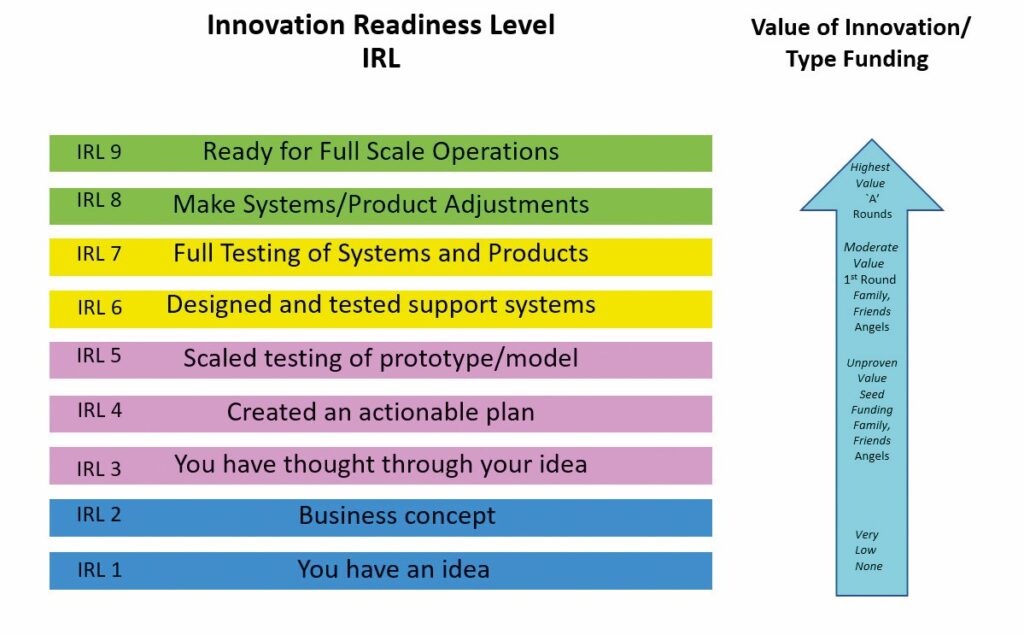
Technical maturity alone does not guarantee real-world adoption. The KTH Innovation Readiness Level (IRL) framework makes this explicit by extending readiness beyond the device into the human, organisational, and economic dimensions. IRL incorporates Customer Readiness Level, Business Readiness Level, Intellectual Property Readiness Level, Team Readiness Level, and Funding Readiness Level. Where TRL asks whether a technology works, IRL asks whether the innovation is understandable, adoptable, financeable, protectable, and operable in its intended context.
The two ladders complement one another. In early stages, customer readiness protects teams from building elegant solutions to the wrong problems. Around TRL 4 and TRL 5, business readiness becomes essential because prototypes, however clever, require a path to affordability, distribution, and service. TRL 6 and TRL 7 align with intellectual property readiness and deeper customer validation so that pilots generate real adoption insights. By TRL 8 and TRL 9, funding readiness and team readiness reduce execution risk by aligning capital and capability with the demands of scale. Each dimension has its own proofs, and gaps among them are early warning signals.

Interdisciplinary work is the quiet engine behind such transitions. Scientists contribute depth on mechanisms. Designers reduce cognitive load and improve usability. Social scientists uncover norms, incentives, and behavioural frictions. Entrepreneurs build models for scale. Policymakers and regulators clarify standards and certification pathways that convert performance into trust. When disciplines work in silos, blind spots multiply and readiness plateaus.
Policy and standards matter concretely at upper readiness levels. In water, national standards often require durability tests, removal efficiencies, and clear operating instructions, while international frameworks shape expectations for safety and quality. In health, diagnostics may need prequalification or approval before procurement, along with evidence that supply chains and training plans exist so devices do not gather dust. These processes take time, but they convert performance into trust, and trust sustains adoption.
THE ROAD TO REAL IMPACT
What distinguishes projects that cross the valley of death from those that do not? Successful teams run technical maturation and market learning in parallel, instead of waiting for a perfect prototype before engaging users. They use stage-gated funding tied to transparent evidence. They measure total cost of ownership, not just sticker price. They choose early markets where the need is strong and switching costs are manageable. They generate decision-grade evidence packs that make it easy for buyers, partners, and regulators to proceed.
Another distinguishing trait is respect for human behaviour. Adoption grows when benefits are clear, frictions are low, rituals are acknowledged, and trust is earned. In our water example, a visible flow indicator or a simple cleaning guide can improve adherence. In diagnostics, a clear readout can shorten training time for health workers. These small design choices accumulate into large differences in adoption.
Research bodies and funders increasingly recognise that readiness is multidimensional. Discussions now emphasise pairing TRLs with agile, responsible mechanisms so that public funding supports not only invention but also translation and uptake. This aligns with practical experience, which shows that innovation succeeds when evidence accumulates across technical, social, and economic dimensions simultaneously. Adoption is sustained by trust. Trust is sustained by proof. And proof must be gathered in the environments where life actually happens.
In the Indian context, where a vibrant higher education system coexists with a rapidly growing startup ecosystem, understanding TRL and IRL frameworks can help academicians, researchers, and students better align their work with real-world needs. For faculty and research supervisors, these frameworks provide structured pathways to design projects that go beyond publications toward technology demonstration and deployment. For students, they clarify how an idea develops—from hypothesis to prototype to field validation—and foster deeper collaboration with industry and user communities. For research institutions and innovation centres, TRL and IRL offer a common language that simplifies evaluating maturity, planning translational milestones, and attracting funding. In short, these frameworks can help Indian science accelerate its shift from knowledge creation to tangible societal impact.
There is also a caution. TRLs can be misused if teams overclaim levels without matching evidence, or if funders treat levels as labels rather than learning tools. The remedy is straightforward. Attach each claim to transparent tests, share protocols openly, invite independent verification, and allow levels to move backwards when new facts emerge. Pre-registering key tests and sharing protocols raises confidence without slowing teams.
The journey from TRL 0 to TRL 9 is never merely technical. It is also ethical and human. A purifier that works only in a laboratory is a technical outcome. A purifier that works in remote schools, clinics, relief camps, and rural homes is a societal outcome. The same is true for diagnostics. It is not enough that a test detects disease. It must reach the clinician’s hand in time to change a decision. Responsible innovation therefore weighs affordability, maintainability, end-of-life handling, and the total cost of ownership over time.
Many technologies that enrich lives today exist because someone stayed steady through uncertainty. Rapid diagnostics, biosand filters, solar lighting systems, satellite-based crop tools, and community water solutions all endured detours and redesigns before gaining trust. They advanced because teams blended scientific discipline with empathy, used readiness frameworks to see blind spots early, and gathered the right evidence at the right time.
When a scientist sees clean drinking water flowing from a device that began as an untested thought at TRL 0, there is quiet fulfilment. Innovation becomes a public good shaped by evidence and care. New ideas will be born tomorrow, once again at TRL 0. With thoughtful readiness and steady collaboration, many will rise to TRL 9 and beyond, touching lives, widening opportunity, and strengthening collective hope.
*Joydeep Dutta is a Professor at KTH Royal Institute of Technology, Stockholm, Sweden, and cofounder of Stockholmwater-technology AB, Caplyzer AB, Kitocoat AB, and HyEnGen AB. Kishore Paknikar is the ANRF Prime Minister Professor at COEP Technological University, Pune, former Director, Agharkar Research Institute, Pune, and Visiting Professor, Indian Institute of Technology Bombay.










